Understanding the importance of buffer solutions in chemistry is crucial for researchers and students alike. Buffer solutions play a critical role in maintaining a stable pH, which is essential in numerous biochemical and industrial processes. This article delves into the top secrets of buffer solutions, providing expert perspectives, practical insights, and evidence-based statements.
The Fundamentals of Buffer Solutions
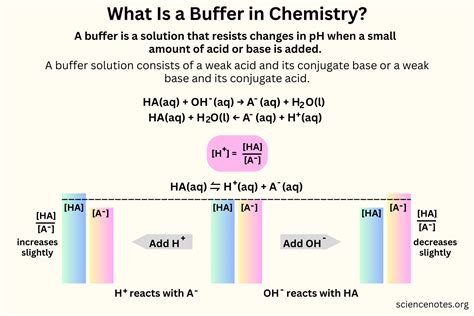
Buffer solutions are composed of a weak acid and its conjugate base or a weak base and its conjugate acid. Their primary function is to resist changes in pH when small amounts of an acid or a base are added. For instance, the bicarbonate buffer system in blood helps maintain the pH within a narrow range, which is vital for physiological processes.Key Insights
Key Insights
- Buffer solutions prevent drastic pH changes, maintaining stability in biochemical environments.
- A critical technical consideration is the buffer’s capacity, which is influenced by the concentrations of the weak acid and its conjugate base.
- A practical application is using a buffer solution to maintain pH during enzymatic reactions, ensuring optimal enzyme activity.
The Chemistry Behind Buffer Solutions
The effectiveness of a buffer solution hinges on the Henderson-Hasselbalch equation: pH = pKa + log([A-]/[HA]), where pKa is the acid dissociation constant, [A-] is the concentration of the conjugate base, and [HA] is the concentration of the weak acid. This equation demonstrates how small changes in the ratio of [A-] to [HA] can significantly affect the pH. For example, in laboratories, phosphate buffers are often used because of their ability to maintain pH around 7, a common physiological pH.Applications in Research and Industry
Buffer solutions are indispensable in various research and industrial settings. In molecular biology, buffers are used in PCR (Polymerase Chain Reaction) to maintain optimal pH levels that support the activity of DNA polymerase. In pharmaceutical manufacturing, buffers are employed to stabilize the pH of active pharmaceutical ingredients, ensuring their efficacy and safety. For example, tris-acetate-EDTA (TAE) buffer is commonly used in gel electrophoresis to maintain pH stability and facilitate the migration of DNA fragments.FAQ Section
What makes a buffer solution effective?
A buffer solution is effective when it can absorb added acids or bases without causing significant pH changes. This is determined by the buffer’s capacity and the concentration of the weak acid and its conjugate base in the solution.
Can buffer solutions be used in non-biological contexts?
Yes, buffer solutions are widely used in non-biological contexts such as industrial processes, where maintaining stable pH levels is crucial for the efficiency and quality of the final product.
Buffer solutions are not just theoretical constructs but are essential tools in both scientific research and industrial applications. Their ability to maintain stable pH levels allows for precise control in environments where pH fluctuations could otherwise lead to unpredictable outcomes. From maintaining pH in biochemical reactions to ensuring the stability of pharmaceutical products, buffer solutions are indispensable for both laboratory and industrial processes.