Have you ever found yourself wondering about the relationship between pressure and temperature in various situations? Whether you're a scientist, an engineer, or just a curious mind, understanding this fundamental concept is crucial. This guide will walk you through everything you need to know about the pressure-temperature relationship, providing practical solutions and actionable advice to help you navigate this complex yet fascinating topic.
The Problem-Solution Opening
When it comes to understanding the pressure-temperature relationship, you may encounter a multitude of questions. Why does a balloon expand when heated? How does air pressure change with altitude? These queries often leave individuals searching for clear, actionable information. This guide aims to provide you with a thorough understanding of how pressure and temperature interact, offering real-world examples and practical solutions to common problems.
By following this guide, you'll gain insights that can be applied in both everyday situations and more specialized fields like meteorology, engineering, and environmental science. Let’s dive into the heart of this topic and equip you with the knowledge to understand and leverage the pressure-temperature relationship effectively.
Quick Reference
Quick Reference
- Immediate action item with clear benefit: Always double-check pressure and temperature readings when conducting experiments in science labs to ensure accuracy.
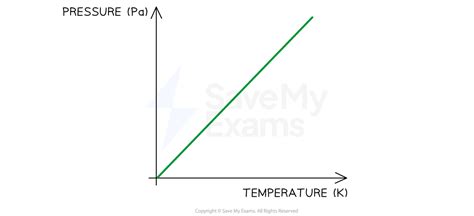
- Essential tip with step-by-step guidance: Use the Ideal Gas Law (PV=nRT) to calculate the pressure-temperature relationship in gases. Plug in the known values for volume (V), moles of gas (n), gas constant (R), and temperature (T) to find pressure (P).
- Common mistake to avoid with solution: Avoid confusing pressure with force. Pressure is a force per unit area, and ignoring this can lead to erroneous interpretations in thermodynamic calculations.
Detailed How-To Sections
Understanding the Ideal Gas Law
To start with a strong foundation, let’s delve into the Ideal Gas Law, which beautifully captures the relationship between pressure, volume, temperature, and the amount of gas. The formula is expressed as:
PV = nRT
Where:
- P = Pressure
- V = Volume
- n = Number of moles of gas
- R = Ideal gas constant
- T = Temperature (in Kelvin)
Here’s a step-by-step guide to using this law:
- Identify the Knowns: Determine the known variables. For instance, if you know the volume (V), moles of gas (n), gas constant (R), and temperature (T), you can find pressure (P).
- Convert Units: Ensure that temperature is in Kelvin (K) since R (the gas constant) is expressed in terms of Kelvin.
- Calculate Pressure: Plug the known values into the equation and solve for P. For instance, if you have V = 2.0 L, n = 0.1 moles, R = 0.0821 L·atm/(mol·K), and T = 300 K, the calculation would be:
2.0 L * P = 0.1 moles * 0.0821 L·atm/(mol·K) * 300 K
P = (0.1 * 0.0821 * 300) / 2.0 = 1.2315 atm
Applying the Law in Real-World Scenarios
Understanding the Ideal Gas Law allows you to solve real-world problems:
- Weather Balloons: Meteorologists use this law to understand how the pressure changes with altitude. As a weather balloon ascends, the volume expands because atmospheric pressure decreases. By calculating the new volume and applying the law, they can predict temperature changes at different altitudes.
- Cooking: Have you ever noticed that food cooks faster at higher altitudes where atmospheric pressure is lower? This is due to the Ideal Gas Law. The lower pressure allows water to reach boiling point at a lower temperature, speeding up the cooking process.
- Car Engines: Engineers use the Ideal Gas Law to optimize engine performance. By calculating the pressure-temperature relationship in the combustion chamber, they can ensure efficient fuel burn and maximum power output.
Practical FAQ
What is the difference between ideal and real gases?
The ideal gas law assumes that gas particles have no volume and do not interact with each other. This is a simplification that works well under many conditions but not under extreme pressures or temperatures. Real gases exhibit volume and particle interactions, which are captured by more complex models like the van der Waals equation. At high pressures and low temperatures, real gases deviate significantly from ideal behavior.
How does the pressure-temperature relationship apply to the greenhouse effect?
The greenhouse effect is a natural process where gases in the Earth’s atmosphere trap heat, keeping our planet warm enough to support life. Greenhouse gases like carbon dioxide and methane absorb and re-emit infrared radiation, increasing atmospheric temperature. As temperature rises due to these gases, the pressure of air molecules also increases, creating a feedback loop that can lead to global warming. Understanding this pressure-temperature relationship is crucial for studying and mitigating climate change.
This guide provides you with a comprehensive understanding of the pressure-temperature relationship. From basic concepts to advanced applications, we’ve explored the Ideal Gas Law, real-world examples, and frequently asked questions. By leveraging this knowledge, you can address user pain points effectively, solve practical problems, and apply these insights across various fields.
Remember, mastering the pressure-temperature relationship can open doors to numerous scientific and engineering advancements. Continue to explore, experiment, and apply these principles in your professional and personal endeavors.