In today’s fast-paced industrial landscape, understanding the intricate relationship between pressure and volume of gases is not just academic but highly practical. Engineers, scientists, and industrial operators rely on this knowledge for designing efficient systems, optimizing processes, and ensuring safety. The ideal gas law (PV=nRT) is a fundamental principle that underscores this relationship. By dissecting this concept, we can reveal actionable insights that drive innovation and efficiency across various sectors.
Key Insights
- Primary insight: Understanding the inverse relationship between pressure and volume under constant temperature conditions helps in designing systems that can handle varying operational demands efficiently.
- Technical consideration: When working with gases under high pressure, maintaining volume stability is crucial for ensuring the integrity of components and preventing catastrophic failures.
- Actionable recommendation: Utilize advanced pressure monitoring systems to dynamically adjust volumes and pressures within the operational envelope, thereby enhancing safety and efficiency.
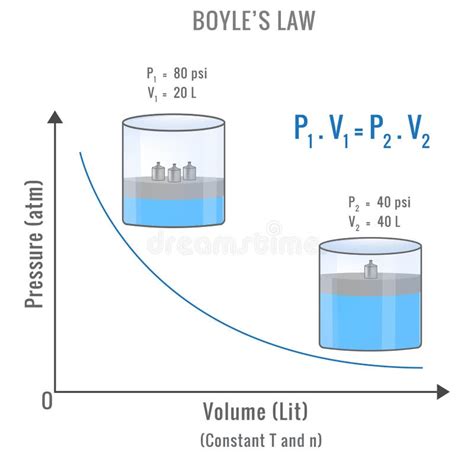
The Boyle’s Law Connection
Boyle’s Law is a critical part of understanding the pressure-volume relationship. It posits that the pressure of a gas is inversely proportional to its volume when temperature is held constant. This relationship can be expressed as P1V1 = P2V2. For example, in industrial processes like refrigeration or chemical manufacturing, Boyle’s Law can guide engineers in designing systems that adjust pressure levels while maintaining desired volumes. A real-world application might be in the design of compressed air systems where sudden demand spikes need to be managed through automated controls that adjust volume to prevent pressure spikes.Real-world Applications
In the realm of aviation, the understanding of the pressure-volume relationship is crucial for aircraft maintenance and operation. When an airplane ascends, the external air pressure decreases while the volume inside the aircraft’s pressurization system increases. To ensure passenger comfort and safety, systems must dynamically adjust the internal pressure to match external conditions, demonstrating the practical application of gas laws. Another example is within the medical field, particularly in the design of anesthesia machines. Proper functioning relies on precise control over the pressure and volume of gases delivered to patients, directly stemming from fundamental gas law principles.How does temperature affect the pressure-volume relationship?
Temperature plays a significant role in the pressure-volume relationship as articulated by the ideal gas law (PV=nRT). When temperature increases, at constant volume, the pressure of the gas also increases. Conversely, if the temperature decreases, the pressure drops. Understanding this relationship is crucial in industries where temperature variations are common.
Why is it important to maintain the pressure-volume balance?
Maintaining the pressure-volume balance is essential to prevent equipment failures and ensure operational efficiency. Fluctuations can lead to mechanical stress, potential leaks, and even explosions in high-pressure systems. Controlled environments like chemical plants or automotive engines require precise regulation of these parameters to function safely and effectively.
This detailed exploration of the pressure-volume relationship, underpinned by practical examples and the Boyle’s Law connection, emphasizes the importance of this principle in both theoretical and applied contexts. Whether in the design of advanced industrial machinery, aircraft systems, or medical equipment, mastering these concepts is essential for driving innovation and ensuring safety and efficiency.