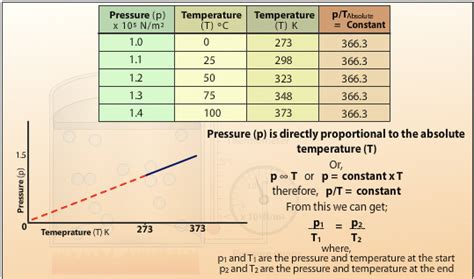
Welcome to our comprehensive guide on understanding the pressure-temperature relationship! If you’re someone who finds complex scientific concepts daunting, don’t worry—this guide will break down everything you need to know in a way that’s easy to understand and practical to apply.
Understanding the Basics
The relationship between pressure and temperature in a gas is governed by several fundamental principles. This guide will focus on making these concepts accessible and actionable, using real-world examples and straightforward advice. By the end of this guide, you’ll have a clear understanding of how pressure and temperature interact and the practical implications of these interactions.
Let's dive right in to address a common issue users face: confusion about the fundamental link between pressure and temperature. This guide aims to clear up any confusion and provide you with actionable insights and practical solutions to apply these principles in your daily life or professional work.
Quick Reference
Quick Reference
- Immediate action item: Record the pressure and temperature of gases around you. This observation can help you understand their relationship.
- Essential tip: Use the Ideal Gas Law (PV=nRT) to predict how changes in temperature or pressure will affect gas volume. This law provides a foundational understanding of the relationship.
- Common mistake to avoid: Assuming that pressure alone affects gas temperature or vice versa. Remember, both pressure and temperature are interconnected through the Ideal Gas Law.
Now that we've got the basics out of the way, let's delve into the details. Understanding the pressure-temperature relationship isn't just about memorizing formulas—it's about applying these concepts to real-world situations.
Detailed How-To Section: Understanding the Ideal Gas Law
The Ideal Gas Law, expressed as PV=nRT, is a cornerstone in understanding the pressure-temperature relationship. Here’s how to use it:
Step 1: Identify Variables
In the Ideal Gas Law, each variable represents an important aspect of gas behavior:
| Variable | Description |
|---|---|
| P | Pressure of the gas, usually measured in atmospheres (atm) or pascals (Pa) |
| V | Volume of the gas, typically measured in liters (L) |
| n | Amount of gas, measured in moles |
| R | Ideal gas constant, value 0.0821 L·atm/(mol·K) or 8.314 J/(mol·K) depending on units |
| T | Temperature of the gas, measured in Kelvin (K) |
Step 2: Apply the Ideal Gas Law
To understand how pressure and temperature are interrelated, consider a simple example:
Imagine you have a balloon filled with gas at room temperature (298 K). If you increase the temperature while keeping the balloon’s volume constant, according to the Ideal Gas Law, the pressure inside the balloon will increase.
Step 3: Calculation Example
Let’s say the initial pressure inside the balloon is 1 atm, the volume is 2 liters, and the temperature is 298 K. If you heat the balloon so the new temperature is 398 K, you can find the new pressure by rearranging the Ideal Gas Law:
Initial state: P₁V₁ = nRT₁
New state: P₂V₂ = nRT₂
Since volume (V) is constant, we can simplify to:
P₁/T₁ = P₂/T₂
Rearrange to solve for P₂:
P₂ = (P₁ × T₂)/T₁
Plugging in the values:
P₂ = (1 atm × 398 K) / 298 K
P₂ ≈ 1.34 atm
Step 4: Real-World Application
Understanding this relationship can be incredibly useful in fields like meteorology, engineering, and even everyday life. For instance, understanding how heating affects the pressure in a closed system can help in designing safer containers for gases.
Detailed How-To Section: Advanced Applications of the Ideal Gas Law
For those who want to take their understanding to the next level, consider how this law applies to more complex scenarios.
Step 1: Mixed Gases and Partial Pressures
When dealing with mixtures of gases, each gas behaves as if it alone occupies the entire volume of the container. Dalton’s Law of Partial Pressures states that the total pressure exerted by a mixture of non-reacting gases is equal to the sum of the partial pressures of each individual gas. Use the Ideal Gas Law to find the partial pressure of each gas.
Step 2: Variable Temperatures
In scenarios where temperature changes over time, you might have to use the Ideal Gas Law in conjunction with other thermodynamic principles. For instance, during heating or cooling processes in a closed system, track changes in temperature and use the law to calculate how pressure will vary.
Step 3: Advanced Calculations
When you need to calculate how pressure changes with volume changes, you’ll apply the Ideal Gas Law iteratively. Suppose you’re dealing with a situation where both volume and temperature are changing. Break it down step-by-step:
1. Calculate initial pressure using the Ideal Gas Law: P₁ = nRT₁/V₁
2. Determine new conditions (volume or temperature changes): Calculate P₂, V₂, or T₂ as needed.
3. Apply the Ideal Gas Law to find the new pressure or volume under new conditions: P₂ = nRT₂/V₂ or V₂ = nRT₂/P₂
Practical FAQ
What is an example of the pressure-temperature relationship in everyday life?
A classic example is the behavior of gases in a tire. When a car is parked in direct sunlight, the temperature inside the tire increases, causing the pressure to rise. This is why tire pressure is often higher in summer than in winter. To avoid tire overpressure, make sure to check and adjust tire pressure regularly, especially during extreme temperature changes.
As we wrap up, it's clear that understanding the pressure-temperature relationship is not only academically interesting but also highly practical. From simple observations to complex calculations, these principles have real-world applications that are crucial in many fields.
Remember, the key to mastering these concepts lies in practicing with real data and scenarios. As you apply the Ideal Gas Law to various situations, you’ll find that this foundational relationship becomes a powerful tool in both your professional and personal life.