Pka and pH relationship lies at the heart of understanding acid-base equilibria in chemistry. The connection between these parameters forms the foundation for various practical applications, ranging from biochemical processes to everyday pharmaceuticals. This article delves into the critical insights linking pKa and pH, providing expert perspectives, evidence-based statements, and real-world examples.
Understanding pKa and pH Fundamentals
The pKa value signifies the acid dissociation constant in a solution, indicating the strength of an acid. It is calculated as the negative logarithm of the acid dissociation constant (Ka). On the other hand, pH measures the hydrogen ion concentration in a solution, indicating its acidity or alkalinity. The relationship between pKa and pH is pivotal for determining the ionization state of acids and bases in various environments.
Critical Insights in the pKa and pH Relationship
Key Insights
- Primary insight with practical relevance: Understanding pKa and pH helps predict how an acid will behave in different solutions, which is essential for optimizing drug formulations and managing chemical reactions.
- Technical consideration with clear application: The Henderson-Hasselbalch equation demonstrates the direct correlation between pKa, pH, and the proportion of ionized and unionized species, providing a powerful tool for chemical engineers and biologists.
- Actionable recommendation: To maximize the efficacy of pharmaceuticals, chemists should calculate the appropriate pH where the drug molecule exists predominantly in its ionized or unionized form.
The Henderson-Hasselbalch Equation: A Practical Approach
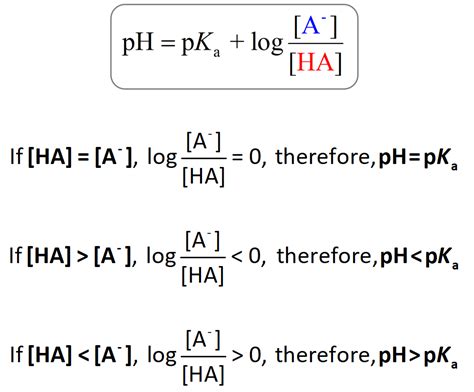
The Henderson-Hasselbalch equation bridges the pKa and pH relationship, offering a practical approach to solving acid-base problems. The equation is given by: pH = pKa + log ([A-]/[HA]). Here, [A-] represents the concentration of the conjugate base, while [HA] is the concentration of the undissociated acid. This equation not only simplifies complex calculations but also aids in understanding the behavior of buffer solutions in biochemical contexts.
Applications in Drug Development and Biotechnology
The pKa and pH relationship is particularly crucial in drug development. The ionization state of a drug molecule can significantly affect its absorption, distribution, metabolism, and excretion (ADME) properties. For instance, an antibiotic like sulfonamide is most effective when it remains in its ionized form to penetrate bacterial cell walls. By manipulating the pH of the gastrointestinal tract, chemists can enhance the absorption rate of such drugs. Understanding these dynamics allows for the precise design of formulations that maximize therapeutic effectiveness while minimizing side effects.
FAQ Section
What is the significance of pKa in pharmaceutical formulations?
The pKa value determines the ionization state of a drug, which influences its solubility, absorption, and overall efficacy. By adjusting the pH of the formulation, the desired proportion of ionized or unionized drug can be achieved to optimize therapeutic outcomes.
How does pH affect the stability of an acid-base solution?
pH affects the stability of acid-base solutions by influencing the ionization equilibrium. At a pH where the acid is predominantly undissociated, it remains stable, whereas in a solution with a pH close to the acid’s pKa, the dissociation increases, potentially affecting stability and reactivity.
By deeply understanding the pKa and pH relationship, professionals in chemistry and related fields can design more effective drugs and optimize chemical reactions, ultimately leading to better outcomes in both industrial and biological applications. This nuanced knowledge underscores the importance of integrating theoretical chemistry principles with practical applications in real-world scenarios.